GENOSS™ DES

Back
Stent for Coronary Arteries
GENOSS™ DES
Description
– Cobalt chromium stent with a thickness of 70 μm.
– Drug coating the minimum required vessel contact.
– Excellent deliverability & fast and thin endothelial process.
More Information
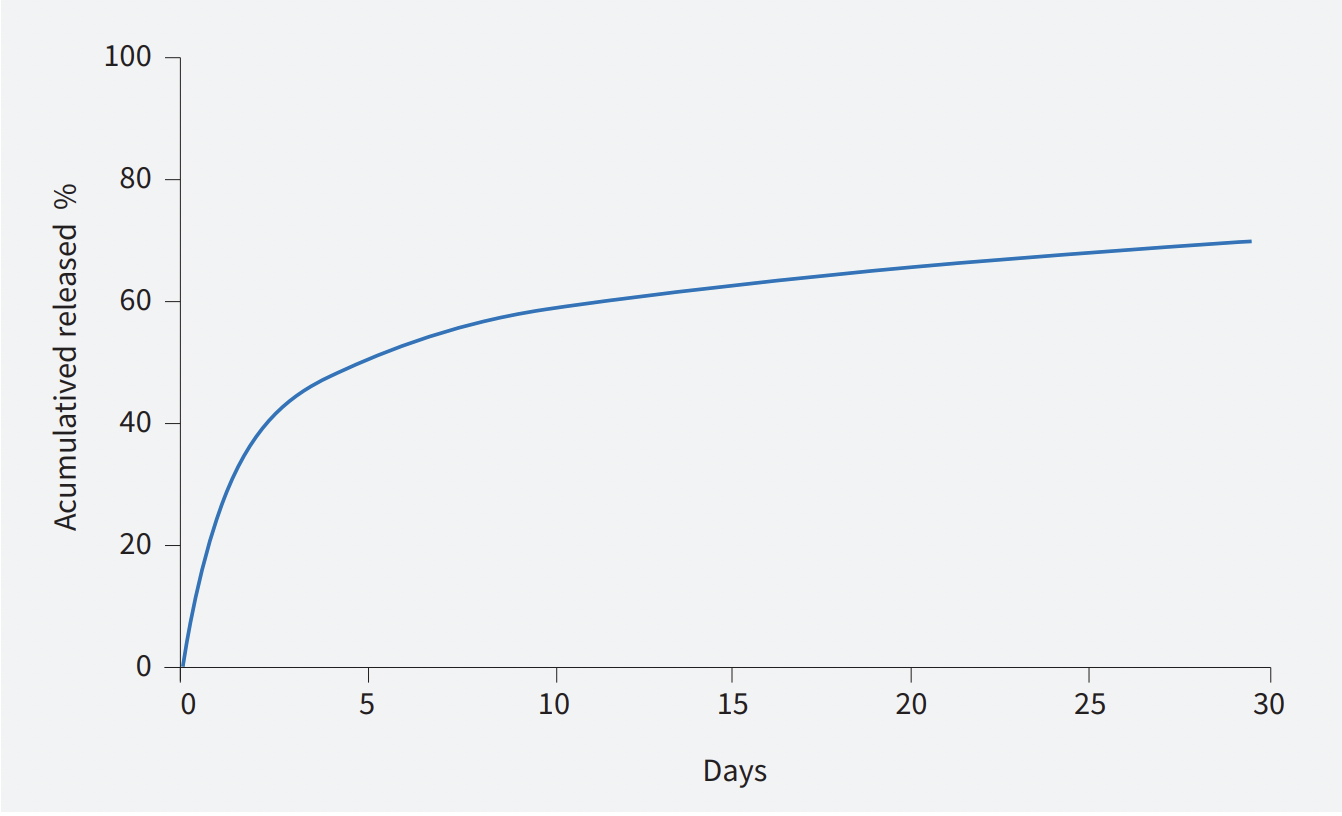
Sirolimus eluting coronary stent
- Control of well-known drug, sirolimus for optimal release
Ultra-thin strut of cobalt-chrome alloy
- 70µm of strut thickness for exceptional flexibility and minimal vessel injury
Abluminal bioresorbable polymer
- Minimized use of coating materials for controllable release rate of drug (coating thickness < 4 µm)
- Limited coating of abluminal bioresorbable polymer on part where stent contacts with the inner wall of vessels
- Transformation into a ‘bare metal stent’ after the release of the drug and termination of degradation of
biocompatible polyme
Improved flexibility
- Great flexibility in complex lesions and effective navigation in tortuous anatomy by open-cell design
- Superior deliverability without compromise in complex anatomy
Optimized radial force
- Resistance to compression of lesion and maintenance of luminal gain.
Low profile delivery catheter system
- An effective system for small vessel treatment with improved trackability
Hydrophilic coating
- A coating with great trackability and crossability
Specification
| Stent material | Cobalt-Chromium alloy, L-605 |
| Polymer | Abluminal bioresorbable coating |
| Drug | Sirolimus |
| Drug dose | 1.02 µg/mm² |
| Stent strut thickness | < 70 µm, < 75 µm |
| Drug coating thickness | < 4 µm |
| Stent strut width | < 90 µm |
| Nominal pressure | 9 atm (2.25 ~ 3.50 mm), 10 atm (3.75 ~ 4.00 mm) |
| Rated burst pressure | 16 atm (2.25 ~ 3.50 mm), 14 atm (3.75 ~ 4.00 mm) |
| Distal shaft coating | Hydrophilic coating |
| Distal shaft | 2.8 Fr (0.92 mm) |
| Proximal shaft | 2.0 Fr (0.68 mm) |
| Lesion entry profile | 0.017″ (0.43 mm) |
| Usable length | 1450 mm |
| Stent shortening | < 2% |
| Stent recoil | < 2% |
| Min. guiding catheter ID | 5 Fr (0.056″) |
| Max. guide wire OD | 0.014″ (0.36 mm) |
| Catheter type | Rapid exchange |
| Shelf life | 2 years from sterilization date |

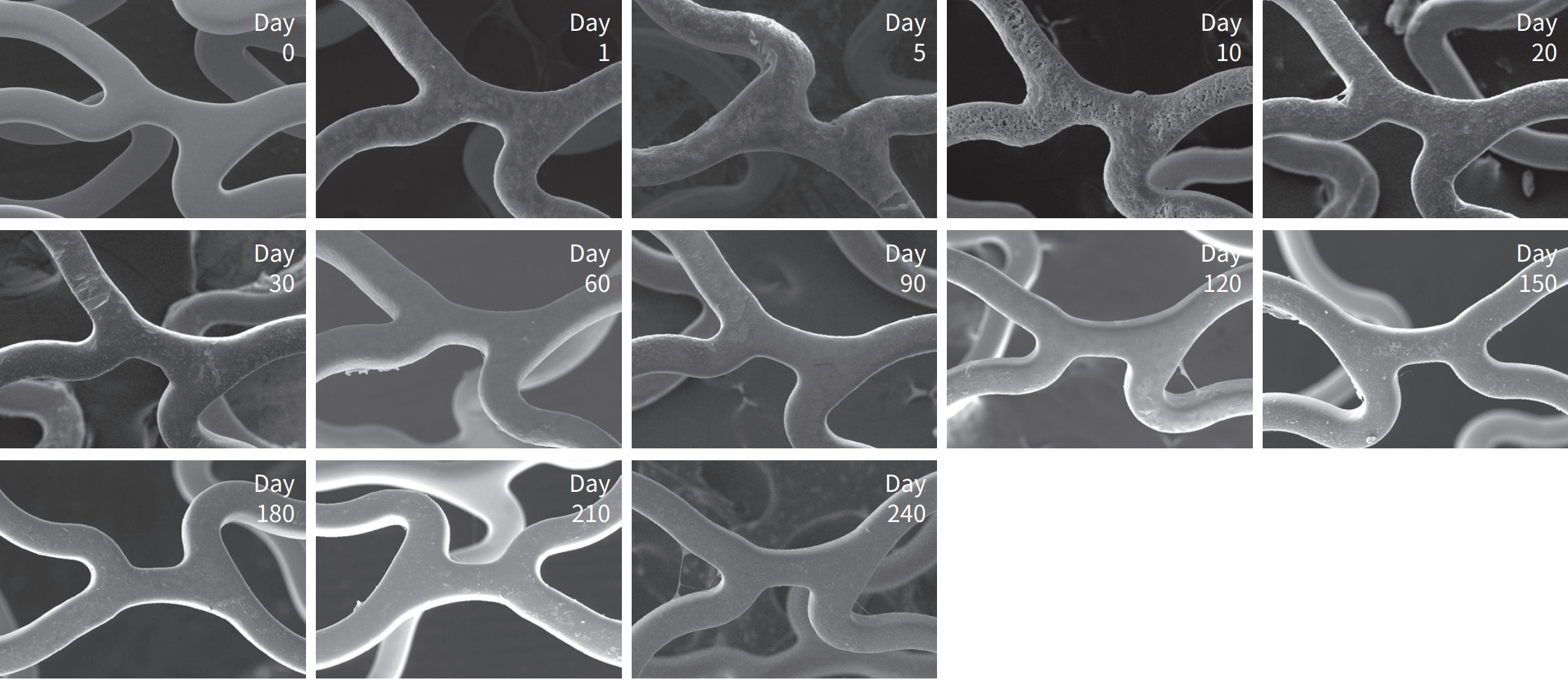
Abluminal resorbable polymer

Polymer coating integrity after expansion
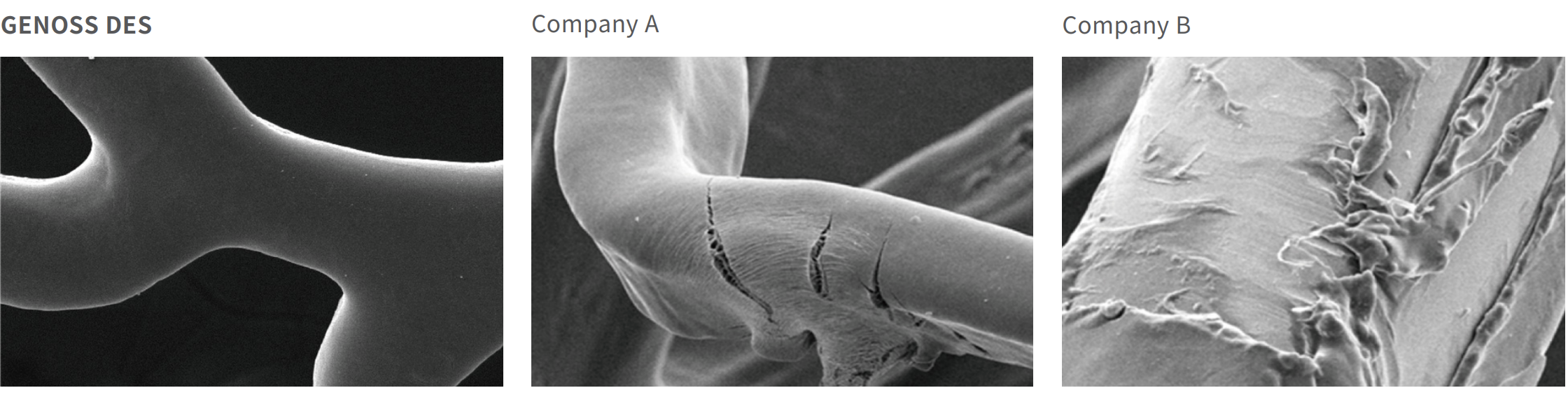
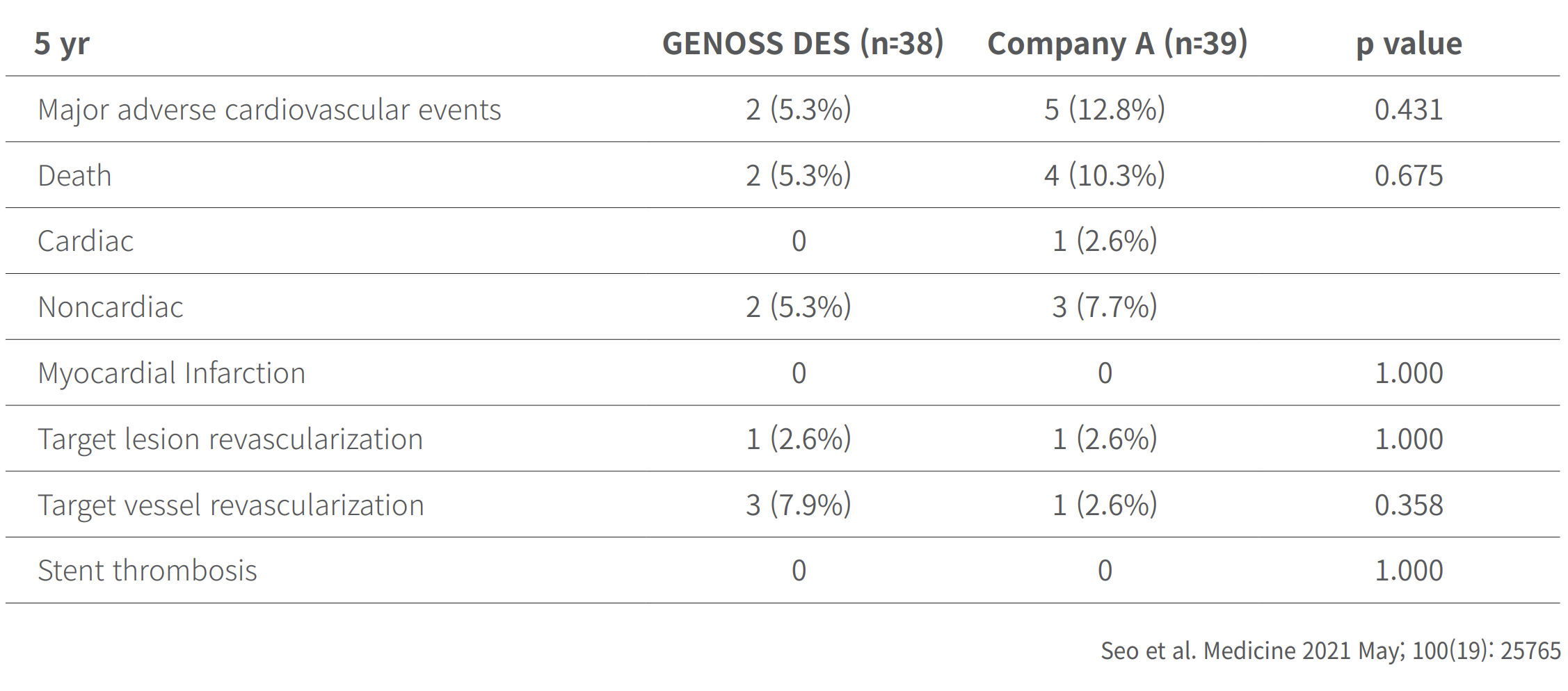
Clinical outcomes at 5 years
Sirolimus Release Profile
Bioresorbable polymer

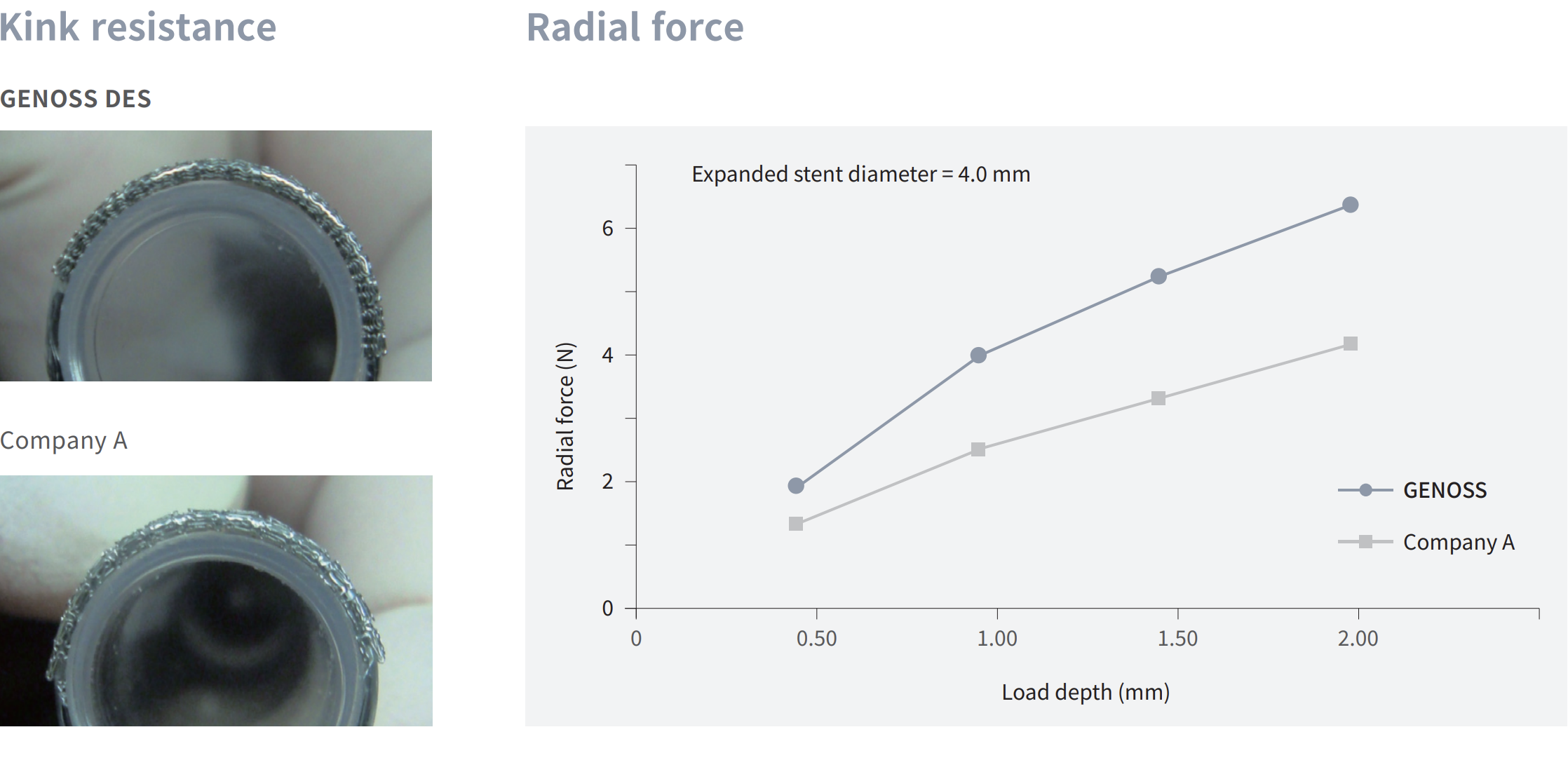
Flexibility

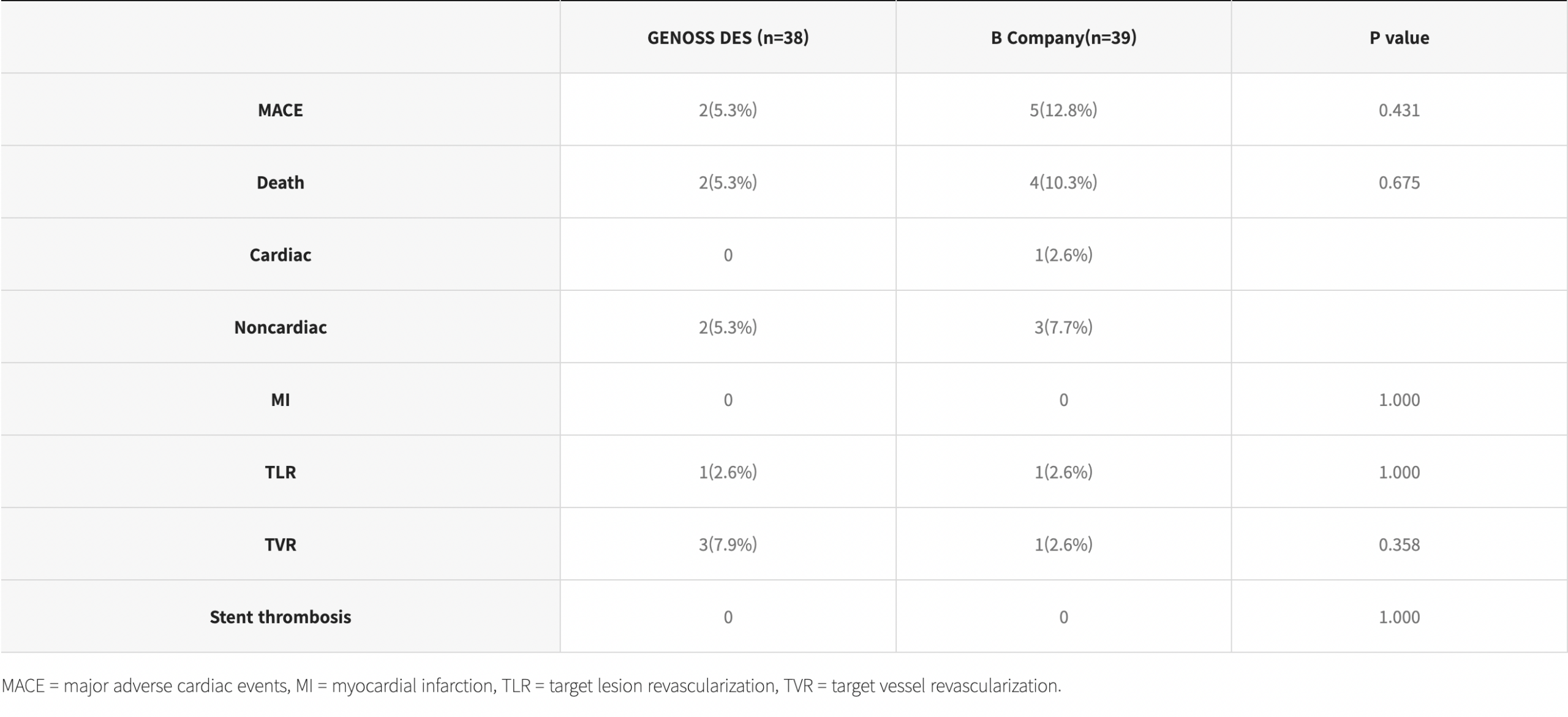
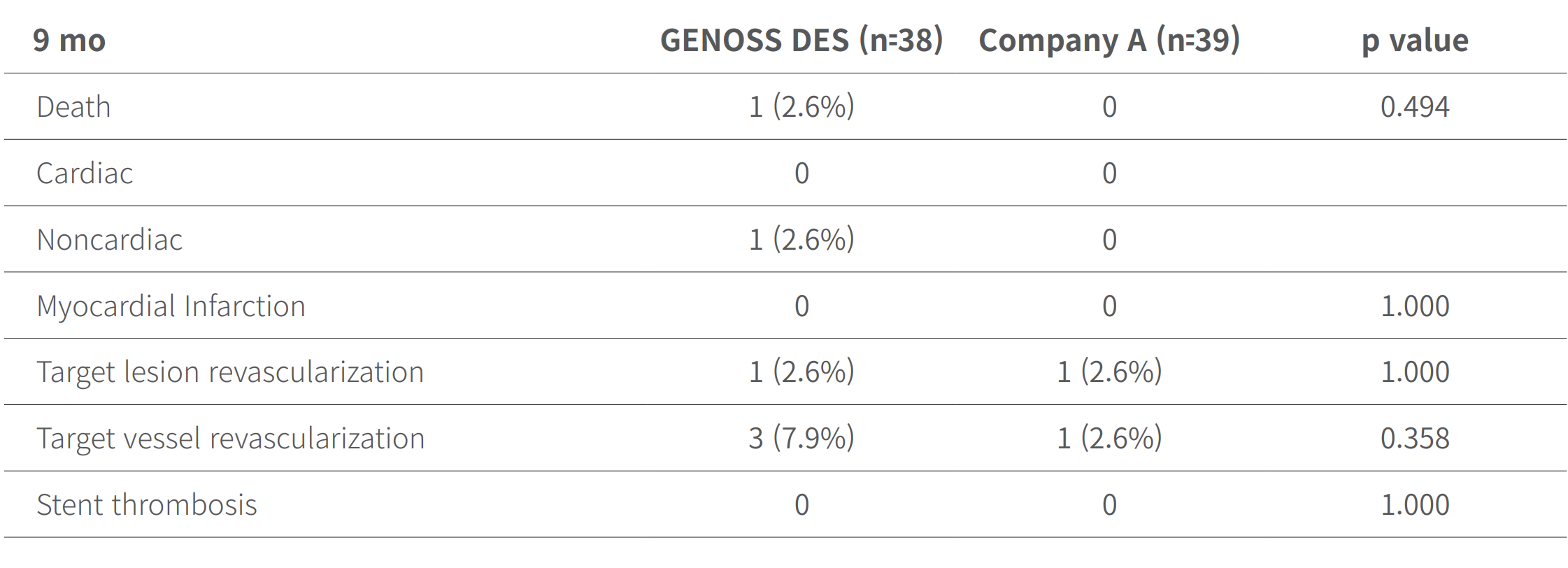
Clinical outcomes at 9 months and at 5 years
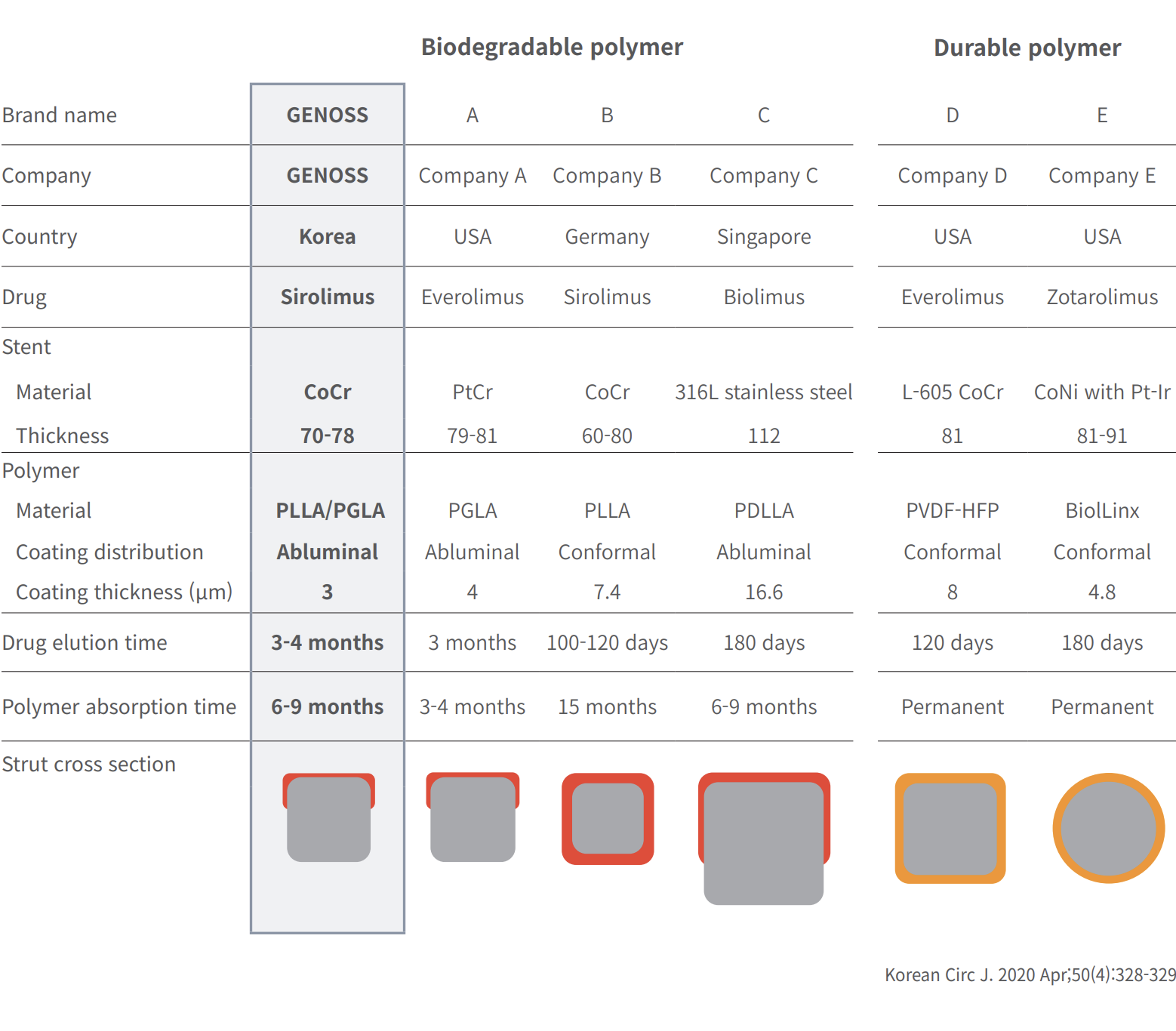
Comparative characteristics of contemporary coronary drug-eluting stents
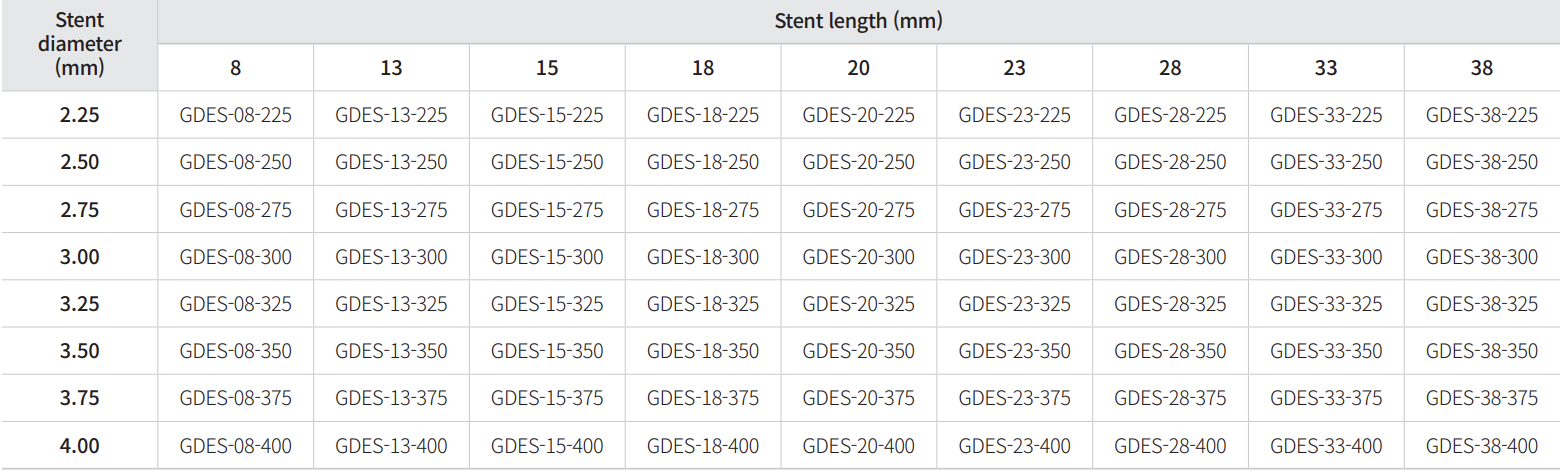
Model Name